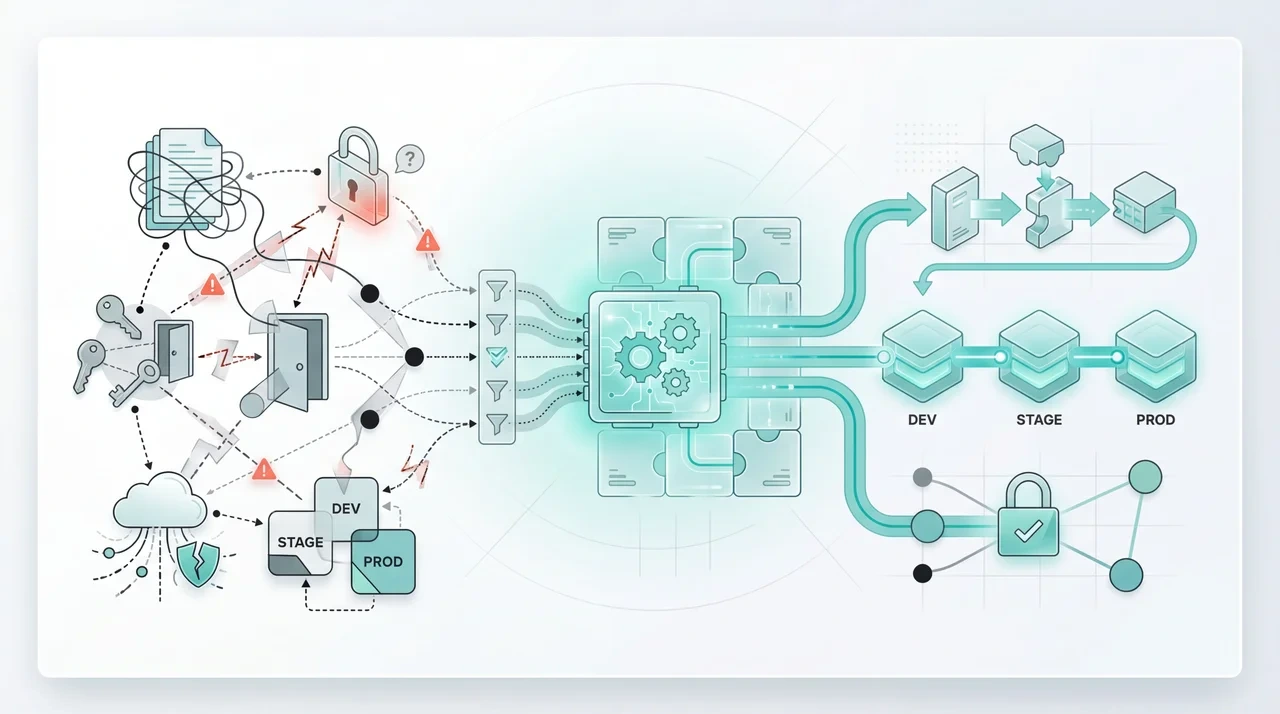
EsperLife’s products are built to improve the practical economics and control points of regulated product delivery.
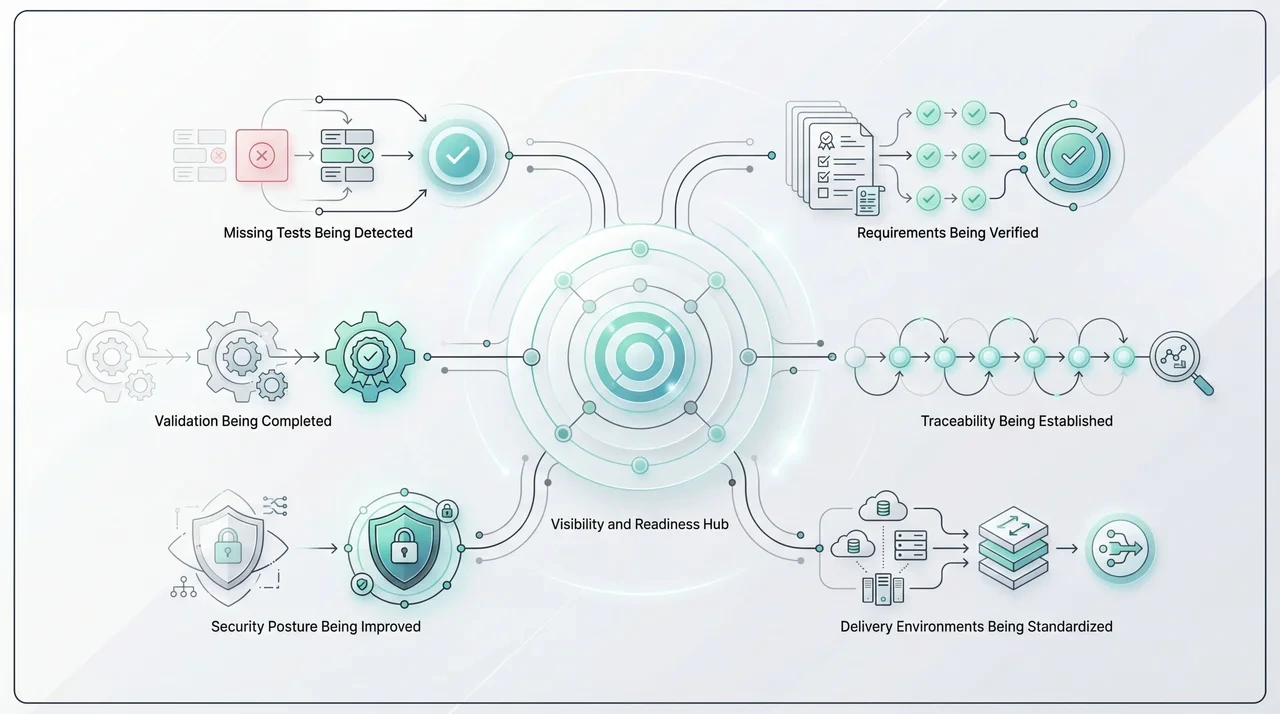
They help reduce the time and cost associated with compliance documentation, validation support, manual traceability maintenance, and repetitive review cycles. They also help teams identify security misconfigurations, access-control issues, and data-governance problems earlier.
By standardizing environments across development, staging, and production, these products support more repeatable execution and reduce avoidable variation in delivery. They also help organizations move product roadmaps forward faster without needing equivalent increases in headcount.
Over time, they create a stronger feedback loop across engineering, validation, compliance, and operations. Historical delivery patterns can be used to suggest architecture improvements, expose test coverage gaps, and identify performance or process issues earlier.